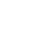Service Hotline
Service Hotline-
ADD:Building 4, Boji Juchuang Technology Park, No. 108 Xishanqiao South Road, Yuhuatai District, Nanjing City
-
TEL:86-25-58578987
-
EMAIL:contact@mightybio.cn
-
PHONE:13701450423, 18705167767

Nanjing Miaodi Biotechnology ELISA kit for typing of S. aureus enterotoxins
This kit uses a sandwich ELISA format: free enterotoxin molecules in samples—including liquid or solid foods and S. aureus culture supernatants—are captured by antibodies coated on a microplate, forming antigen-antibody complexes. These complexes then react with enzyme-labeled detection antibodies, and the addition of substrate produces a colorimetric signal (Fig. 1). Absorbance is measured at 450 nm using a microplate reader, and the toxin concentration in the sample is directly proportional to the measured absorbance.
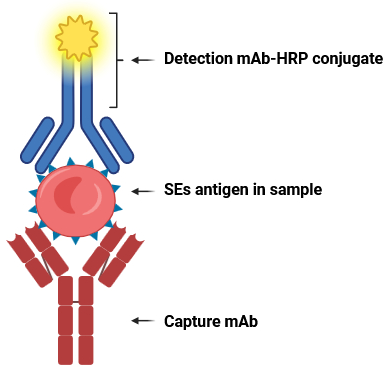
Fig. 1. Schematic diagram of the ELISA system for typing of S. aureus enterotoxins
This kit is designed for 12 assays, allowing simultaneous detection of five staphylococcal enterotoxins (SEA–SEE) in each sample (Fig. 2). Compared with similar domestic and international products, this kit demonstrates excellent specificity and sensitivity, with no cross-reactivity among the five toxins (see Tables 2–4). The detection limit reaches 0.25 ng/mL (ppb) (see Table 1).
Unlike some kits that use biotin-streptavidin amplification systems, this kit produces very low background and completes the assay in less than 2 hours (excluding sample pretreatment).
Moreover, all antibodies used in this kit are produced in-house rather than commercially sourced, minimizing batch-to-batch variation and ensuring stable, reliable quality. The calibrators are naturally derived native enterotoxins (SEA–SEE), not recombinant antigens, ensuring high fidelity and accuracy of the assay.

Fig. 2. Schematic diagram of the toxin types detected by the S. aureus enterotoxin typing ELISA kit.
Supplementary data
Table 1. Detection limits of the S. aureus enterotoxin typing ELISA kit in various food matrices
Toxin Matrix | SEA (ng/ml) | SEB (ng/ml) | SEC (ng/ml) | SED (ng/ml) | SEE (ng/ml) |
PBS | 0.25 | 0.25 | 0.0625 | 0.0625 | 0.25 |
Milk# | 0.25 | 0.25 | 0.0625 (0.0625) | 0.0625 | 0.25 |
Milk Powder※ | 0.25 | 0.25 | 0.0625* | 0.0625 | 0.25 |
Rice※ | 0.25 | 0.25 | 0.0625** | 0.0625 | 0.25 |
Noodles※ | 0.25 | 0.25 | 0.0625*** | 0.0625 | 0.25 |
Tofu※ | 0.25 | 0.5 | 0.0625 | 0.0625 | 0.25 |
Fermented Milk※ | 0.25 | 1.0 | 0.5 | 0.0625 | 0.25 |
Ice Cream※ | 0.5 | 0.25 | 0.125 | 0.125 | 0.25 |
Marinated Beef※ | 0.5 | 0.25 | 0.03125 | 0.0625 | 0.125 |
BHI Broth | 0.25 | 0.25 | 0.0625 | 0.0625 | 0.25 |
# Detection result after weighing 1 g of milk powder and adding 10 mL of PBS.
※ Detection result after weighing 10 g of food matrix and adding 15 mL of PBS.
Table 2. Detection results of the Staphylococcus aureus enterotoxin typing ELISA kit in culture supernatants of S. aureus strains (20 strains in total.
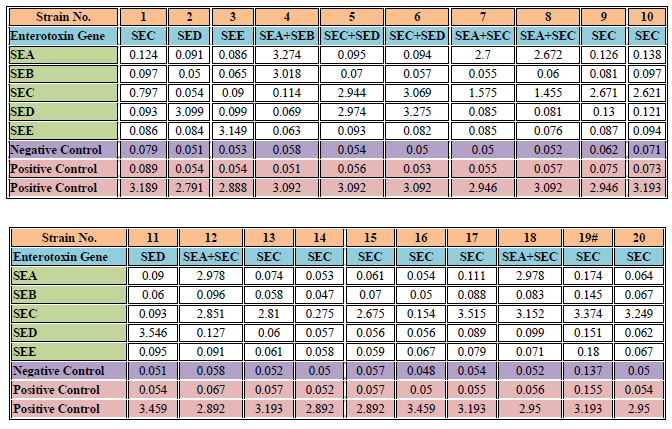
The OD value of the negative control well for the culture supernatant of strain No. 19 was > 0.1.
Table 3. Detection results of culture supernatant from S. aureus strain No. 19 after dilution with PBS
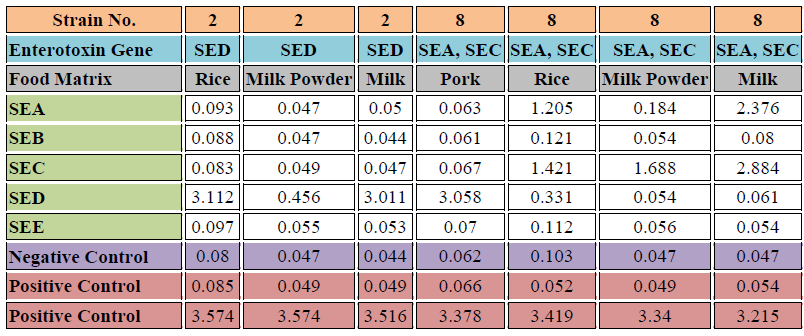
Table 4. Detection results of S. aureus strains (Nos. 2 and 8) spiked into food matrices #
Strain No. | 19 | 19 | 19 |
Enterotoxin Gene | SEC | SEC | SEC |
Dilution | Undiluted | 1:5 | 1:10 |
SEA | 0.175 | 0.097 | 0.088 |
SEB | 0.099 | 0.068 | 0.060 |
SEC | 2.381 | 2.544 | 2.886 |
SED | 0.161 | 0.093 | 0.077 |
SEE | 0.165 | 0.078 | 0.073 |
Negative Control | 0.135 | 0.054 | 0.049 |
Positive Control | 0.159 | 0.054 | 0.054 |
Positive Control | 3.023 | 3.122 | 3.084 |
# S. aureus overnight culture was centrifuged, the supernatant was discarded, and the cell pellet was resuspended in PBS. The bacterial cells were then spiked into the food matrix and incubated at room temperature for 36 hours. Sample pretreatment was performed according to the kit instructions.
Product Catalog No.: ES24006